
Objectives: ![]()
What is the purpose of the work?
In this proposal, we take advantage of very recent developments made in our research group on cellulose surface modification using selective oxidation reactions to design new techniques for surface immobilization of biomolecules, particularly hIgG. Therefore, the best case scenario to minimize the loss of proteine activity and to overcome the poor retention is by selecting a proper immobilization technique, supporting selection and optimum immobilization conditions, in order to preserve the proteine activity as close as to its original pefomrance level by not changing the chemical nature or reactive groups in the proteine binding site (Figure 1).
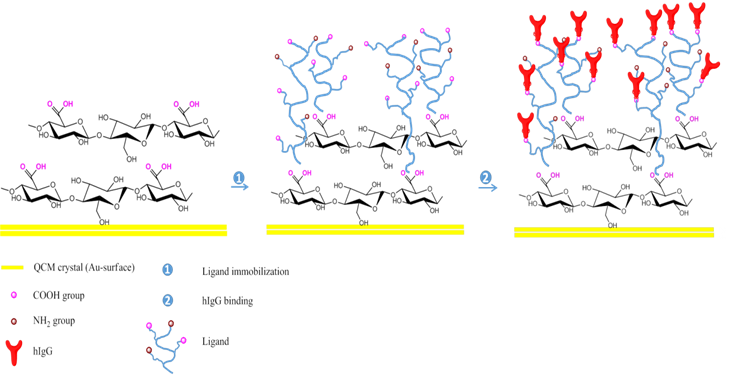
Figure 1. Schematic illustration of the preparation oxidized cellulose-ligand biointerface for hIgG detection.
2017
O1. Creating a highly nanostructured oxidized cellulose surface
2018
O2. Conjugation of binding sites (ligands) on activated oxidized cellulose surfaces monitored in-situ by QCM-D and SPR
2019
O3. Detection and binding of the protein (immunoglobulin G) on ligands supported polymer biointerface
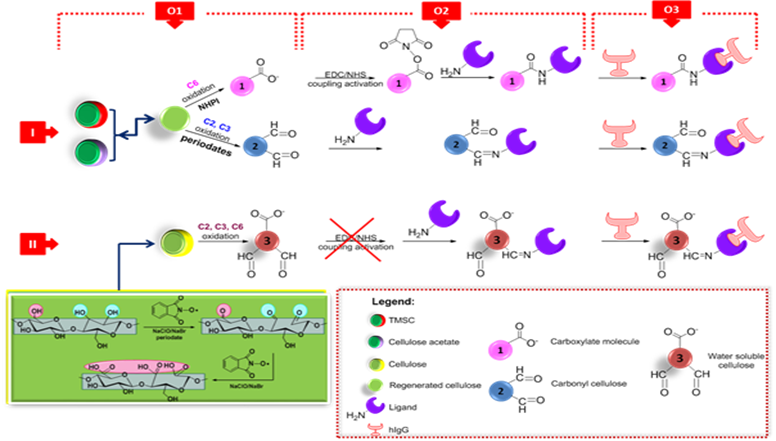
Figure 2. The general schematization of the proposed approach for the detection and binding of the protein (immunoglobulin G) on ligands supported polymer biointerface.